Twin to twin transfusion syndrome
This information brochure has been developed by Mater’s Centre for Maternal Fetal Medicine. It is not intended to take the place of comprehensive counseling and support, which is provided as part of our service, but it will provide some background information for you to consider.
What is twin to twin transfusion syndrome (TTTS) and what causes it?
Twin to twin transfusion syndrome is a condition that affects approximately 10 per cent (one in ten) of identical twins that share a single placenta. When twins share a single placenta we call them monochorionic. Usually each twin will have its own sac of amniotic fluid and its own umbilical cord connecting it to the placenta.
Blood vessels that connect the two umbilical cords on the surface of the placenta allow blood from one twin (the donor) to flow into the other twin (the recipient). This transfusion of blood occurs when there is an imbalance of blood flow from the donor twin to the recipient twin which causes twin to twin transfusion syndrome.
How can TTTS affect twins?
 The donor twin has less blood so their urine output decreases causing a reduced amount of amniotic fluid in its sac. The donor twin's bladder can become empty and difficult to see on ultrasound. Reduced amniotic fluid around the donor twin is called oligohydramnios. Sometimes the membrane of the donor sac can be hard to see on the ultrasound due to it covering the baby like cling wrap.
The donor twin has less blood so their urine output decreases causing a reduced amount of amniotic fluid in its sac. The donor twin's bladder can become empty and difficult to see on ultrasound. Reduced amniotic fluid around the donor twin is called oligohydramnios. Sometimes the membrane of the donor sac can be hard to see on the ultrasound due to it covering the baby like cling wrap.
The recipient twin experiences the opposite effect. Having more blood causes the recipient twin to pass large volumes of urine that fills its amniotic sac up like a large balloon. Increased amniotic fluid around the recipient twin is called polyhydramnios and this can lead to the waters breaking (ruptured membranes) and/or preterm birth.
As TTTS progresses, the effects on each twin can become more severe. The recipient twin can have difficulty pumping the extra blood and it may develop signs of heart failure.
If the recipient twin develops heart failure, other parts of its body may become swollen with fluid—this is a life-threatening condition called hydrops. TTTS can alter the blood flow in the umbilical cord of either twin and this can be measured with ultrasound. Due to the nature of the connected blood vessels with TTTS, sometimes one or both twins can die without warning. If one twin dies, the other twin may die as a consequence or it may suffer brain damage and major disability.
How is TTTS diagnosed?
Signs and symptoms of TTTS usually develop mid-pregnancy, between 16 and 26 weeks. The mother may initially notice that her abdomen has become very large and uncomfortable with associated back pain. She may also experience uterine contractions and decreased fetal movements. An ultrasound scan is required to measure the reduced fluid around the donor twin and the increased fluid around the recipient twin for a diagnosis of TTTS.
If TTTS is suspected on an ultrasound scan the mother should be referred to a tertiary facility that is experienced in the accurate assessment and management of TTTS.
What are the stages of TTTS?
A staging system for TTTS based on the ultrasound findings has been developed to help counsel parents and to guide treatment options.
|
Stage one (I)
|
Reduced or no amniotic fluid seen around the donor twin and increased amniotic fluid around the recipient twin
|
|
Stage two (II)
|
An empty bladder in the donor twin
|
|
Stage three (III)
|
Abnormal blood flows in the umbilical cord of either twin
|
|
Stage four (IV)
|
Hydrops of the recipient twin
|
|
Stage five (V)
|
Death of either twin
|
In addition to confirming the diagnosis and stage of TTTS, a thorough ultrasound is undertaken which includes measuring the babies, looking for major malformations and assessing the placental location and cord insertions. A vaginal ultrasound assessment of the cervix is usually performed.
What is selective fetal growth restriction (sFGR)?
When identical twins share a single placenta, they may not share it equally and one twin may fail to grow properly—referred to as growth restriction. The twin with growth restriction will usually be significantly smaller than its co-twin and we call this selective fetal growth restriction.
If the growth restriction is particularly severe it can pose significant risks for both twins. In pregnancies with TTTS, selective fetal growth restriction can occur at the same time, adding extra risk to the pregnancy and making management even more complicated.
What are the treatment options for TTTS?
1. No treatment
Leaving severe TTTS untreated is associated with a high loss rate of both babies, of more than 80 per cent. This can result from prematurity due to the waters breaking and/or early delivery or loss can occur before birth from the direct effects of TTTS.
2. Expectant management or observation
Expectant management involves monitoring the pregnancy by regular ultrasound scans but without any specific treatment. Expectant management is usually recommended for Stage one TTTS, as in some cases it will remain stable or resolve without treatment.
3. Amnioreduction (amniodrainage or amniocentesis)
Amnioreduction is also called amniodrainage or amniocentesis. It is an ultrasound guided procedure that removes amniotic fluid from around the recipient twin with the aim to reduce the risk of the waters breaking and/or early birth. A needle is inserted into the amniotic sac of the recipient twin and one to four litres of amniotic fluid may be removed. An amnioreduction may need to be repeated on more than one occasion during the pregnancy as the fluid can return within a week or two. There is a small risk of the waters breaking and/or early delivery associated with the procedure. Figures from Mater's Centre for Maternal Fetal Medicine have shown that repeated amnioreduction for TTTS results in a survival rate of 60 per cent of babies. Of the survivors, 20 to 25 per cent may have subsequent brain damage and disability.
4. Fetal laser surgery
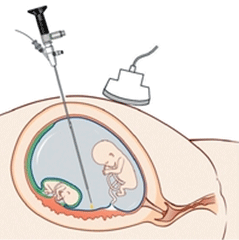 Fetal laser surgery involves passing a small camera (called a fetoscope) into the amniotic sac of the recipient twin. A laser fibre is then used to block the connecting vessels on the surface of the placenta and stop the flow of blood from one twin to the other. Fetal laser surgery is performed in an operating theatre under local anaesthetic and sedation. An overnight hospital stay is required after fetal laser surgery.
Fetal laser surgery involves passing a small camera (called a fetoscope) into the amniotic sac of the recipient twin. A laser fibre is then used to block the connecting vessels on the surface of the placenta and stop the flow of blood from one twin to the other. Fetal laser surgery is performed in an operating theatre under local anaesthetic and sedation. An overnight hospital stay is required after fetal laser surgery.
The main advantage of laser therapy over repeated amnioreduction is that it treats the underlying cause of TTTS, and usually requires only one intervention. It also means that the co-twin is better protected if one twin dies.
This first fetal laser surgery for TTTS in Australia was performed at Mater Mothers' Hospital in 2002. Mater's Centre for Maternal Fetal Medicine continues today as one of the busiest fetal therapy centres in Australasia. The overall survival rate of one or both babies after fetal laser surgery is approximately 75 per cent. The incidence of cerebral palsy or significant developmental delay is up to 10 per cent. The long term outcomes following fetal laser surgery are recognised to be significantly better than those after amnioreduction. The risks of fetal laser surgery include the waters breaking and/or early delivery which occurs in around 15 per cent of cases before 28 weeks. Complications affecting the mother are uncommon, but include breathing problems, bleeding, blood clots forming in the legs and infection.
Cerebral palsy and brain damage may not be prevented by fetal laser surgery, as an injury may have occurred prior to treatment. Fetal death affecting one or both of the twins may occur within the first week following laser treatment. This may reflect an abrupt change in the shared fetal circulation.
Repeat fetal laser surgery is uncommon but may it be required if there is evidence of ongoing harmful blood vessel connections between the twins after an initial treatment.
What is TAPS?
TAPS stand for 'Twin Anaemia Polycythainia Sequence'.
It occurs when one twin has a low blood count (anaemia) and the other twin a high blood count (polycythaemia). It can occur on its own or may happen after laser therapy for TTTS. Some cases of TAPS do not require specific treatment other than regular ultrasound monitoring.
Further information and support
Families affected by TTTS will often experience different feelings and may have many questions related to this condition. In addition to the support offered through Mater's Centre for Maternal Fetal Medicine, there are several support groups available which focus on this condition.
You may also find the following websites useful:
For some parents, both babies survive, while others have gone through the trauma of losing one or both of their babies. Many of the families who have been treated at Mater are happy to speak to other parents facing similar challenges. If you would like to speak to other parents who have had a pregnancy affected by TTTS please let us know.
Social work and pastoral care services
Mater's social work and pastoral care services are available to support our women and families. Please speak to your health care team if you and your family would like to talk to someone from these services.
Referrals
A referral from a doctor is required to access services provided by the Mater Centre for Maternal Fetal Medicine. If you have private health insurance, you may choose to be treated as a private patient. Further information regarding admission procedure and other hospital stay details will be provided at your first appointment.
Costs
Not all services provided by the Centre are covered by Medicare or your private health insurance. A gap payment will be incurred for some services. Bulk billing is available for women with pension or health care cards who present their cards on the day of their service. If you have any queries regarding fees, please discuss them with our staff prior to your visit by phoning 07 3163 1896.
Women traveling to Mater's Centre for Maternal Fetal Medicine for assessment, when their referring hospital is greater than 50 kilometres away, may be eligible to have some of their expenses reimbursed by their referring hospital under the Patient Travel Scheme (PTS). Further information is available via Queensland and NSW Health department websites or by contacting your local hospital’s travel coordinator.
Accommodation
Reasonably priced short-term accommodation, including motels, Ronald McDonald House and Mater's Reg Leonard House are within walking distance of Mater. Our travel coordinator can assist you with organising affordable, local accommodation.
Parking and transport
Paid parking is available on site at the Hancock Street Car Park and Mater Hill car parks. Parking charges apply.
Mater Hill Busway station is situated in the middle of Mater’s South Brisbane campus. You can access bus timetables at translink.com.au/.
Research
The Mater Centre for Maternal Fetal Medicine has a strong history of research to improve outcomes in high risk pregnancies. Your participation in any research at the Mater is entirely voluntary.
Mater Foundation
The success of our fetal therapy program has been built on the support of businesses and the public through donated funds. Any donations will be gratefully received. All donations are tax deductible and should be made payable to the Mater Foundation, 580 Stanley Street, South Brisbane Qld 4101. For further information please phone 07 3163 8000.
Contacts
For further information please contact the Centre between 8.30 am and 4.30 pm, Monday to Friday by phoning 07 3163 1896.
Alternatively you may contact the Centre by email at mfm@mater.org.au
If required your doctor can contact a Maternal Fetal Medicine Specialist directly by phoning 07 3163 8111.
Centre for Maternal Fetal Medicine
Level 7
Mater Mothers’ Hospital
Raymond Terrace
South Brisbane Qld 4101
Phone: 07 3163 1896
Fax: 07 3163 1890
Mater acknowledges consumer consultation in the development of this patient information.
Mater Doc Num: PI-CLN-430149
Last modified 19/10/2017.
Consumers were consulted in the development of this patient information.
Last consumer engagement date: 19/1/2014
For further translated health information, you can visit healthtranslations.vic.gov.au/ supported by the Victorian Department of Health and Human Services that offers a range of patient information in multiple languages.